FLEX-FIX Syndesmosis Fixation Device
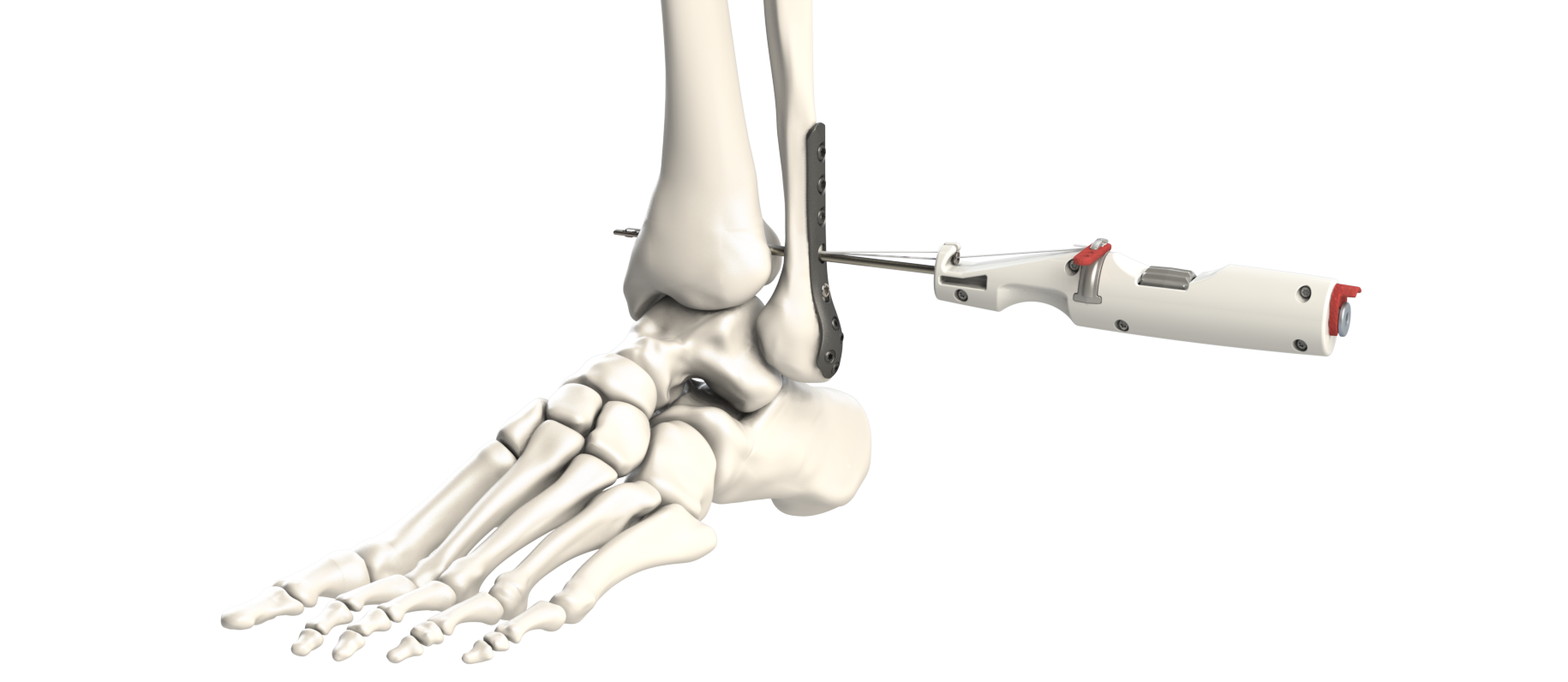
The Flex-Fix was my baby. Syndesmosis injuries (high ankle sprains involving the ligament connecting the tibia and fibula) need stable fixation to heal properly. I designed the complete system: a delivery instrument, a small toggle anchor and button connected by suture, all packaged as a sterile single-use kit.
This project took me all the way to Taiwan to work directly with our contract manufacturer — training their team on cleanroom assembly procedures for the finished product. We then packaged the final labeled assembly in-house in our own clean room. I drove the entire project from design and prototyping through first-article production, mechanical testing, documentation, patent application, and 510(k) submission. The FDA clearance came through in July 2024 — by then I'd already transitioned to Truckhouse full-time, but the work was done. End-to-end ownership in every sense.
Complete Delivery System
Purpose-built delivery instrument, toggle anchor, button, suture assembly, drill bit, and washer for solo-use (without a distal fibula plate).
Sterile-Packaged
Unlike tray-based products, this was designed as a single-use sterile kit with different manufacturing and packaging challenges.
Toggle Anchor Mechanism
A thumb-wheel causes an internal shaft to translate, pushing the toggle-button out of the tip of the handle. Tension in the sutures causes the toggle-button to rotate into position — a novel use of rotation to gain mechanical advantage, making deployment easy and predictable.
International Supplier Management
Traveled to Taiwan to train the contract manufacturers on cleanroom assembly processes and meet their contracted suppliers for non-metallic parts of the assembly — injection molded plastic and silicone components.
End-to-End Ownership
Concept, detailed design, prototyping, V&V, regulatory submission, supplier qualification, manufacturing transfer, marketing materials and surgical technique guides, and recurring meetings with friendly surgeons to ensure product success.
This was a solo design effort from concept through commercialization.
FDA 510(k) cleared and commercially launched. As a solo engineer, I owned every phase — injection molded design, sterile packaging, cleanroom assembly, verification and validation, patent navigation, and international supplier training.
